
111, How many neutrons does the isotope 19980Hg have? 10.6514 amu \\C. Which functional group is represented in the diagram below? Which one of the following pairs are isotopes? View the full answer Transcribed image text: of the following represent isotopes? 2003-2023 Chegg Inc. The isotopic abundance and atomic weights vary in normal materials, but upper and lower bounds of the standard atomic weight have not been assigned by IUPAC or the variations may be too small to affect the standard atomic weight value significantly. Radon-220, Polonium-210, Platinum-195 Express answer as an isotope, Which of the following represent isotopes? Which of the following isotopes is more likely to decay via beta particle (electron) emission than by positron emission? Both 40K and 40Ca have the same atomic number (proton number), which is 19 for potassium and calcium.However, they differ in the number of neutrons, making them isotopes of the same element. Element has two ore more isotopes that are used to determine its standard atomic weight. Which of the following pairs represent isotopes? Which of the following is true about the relative abundances of 12X and 13X? The experimental rate law of the. a) Atomic mass b) Chemical stability c) Number of neutrons d) Number of protons, The element X has an atomic mass of 12.80 amu. erties of salts? A: The given data contains, These are isotopes of hydrogen and hence, each of them contain 1 proton but the number of neutrons are different. Write the atomic symbol (^A _Z X) for each of the isotopes described below. A) Ca^ 2 and Mg^ 2 B) ^199Hg and ^200Hg C) ^180O and ^19F D) ^238U and ^238Np, Which of the following isotopes is most likely to be unstable? (a) Write the atomic symbol for each of these atoms. Write the isotopic symbol for the sulfur isotope with 16 neutrons. H, H a) Bromine b) Aluminium c) Polonium d) Carbon, How many neutrons does the isotope 7734Se have? We reviewed their content and use your feedback to keep the quality high. Mass of proton = 1.0073 amu, A: Neutral atom are those having not any charge. A: Following equation will be used to solve this equation. 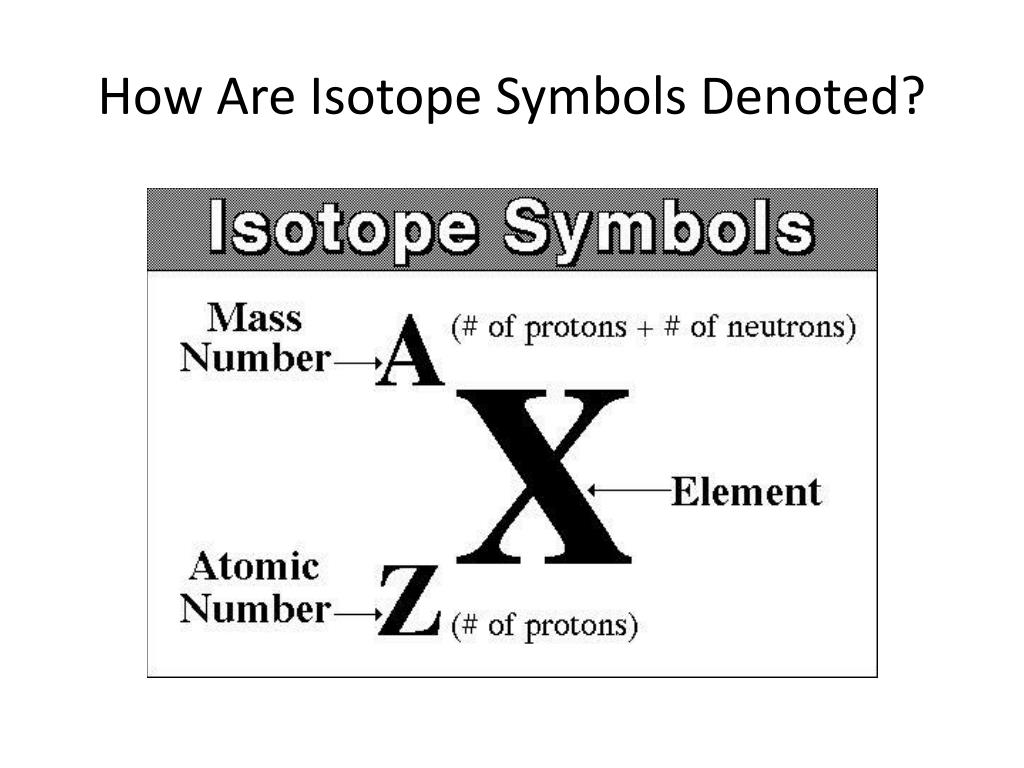
Select one: You perform the following chemical reaction by mixing a piece of magne A. a) 14C b) 12C c) 2H d) 16O, Identify the isotope that has 56 protons and 84 neutrons. Unlock a special one-week offer to get access to this answer and millions more. Khareedo DN Pro and dekho sari videos bina kisi ad ki rukaavat ke! An element with two isotopes has a tabulated atomic mass of 161.71 u. represent an isotope, the mass number is written next to the symbol on the top left. The atomic mass (A) is obtained by adding the number of protons and neutrons in a given nucleus of a chemical element. Any given element can have more than one isotope. These atoms have the same number of protons but different mass numbers. Calculate the atomic mass of silicon given the following data for its three natural isotopes. Which of the following represents a pair of isotopes? Silicon is the second most abundant element in Earths crust.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
